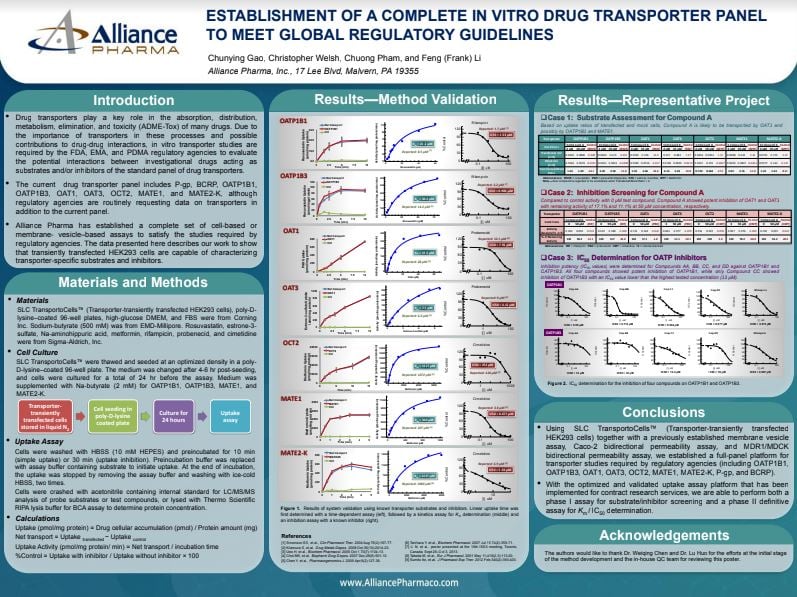
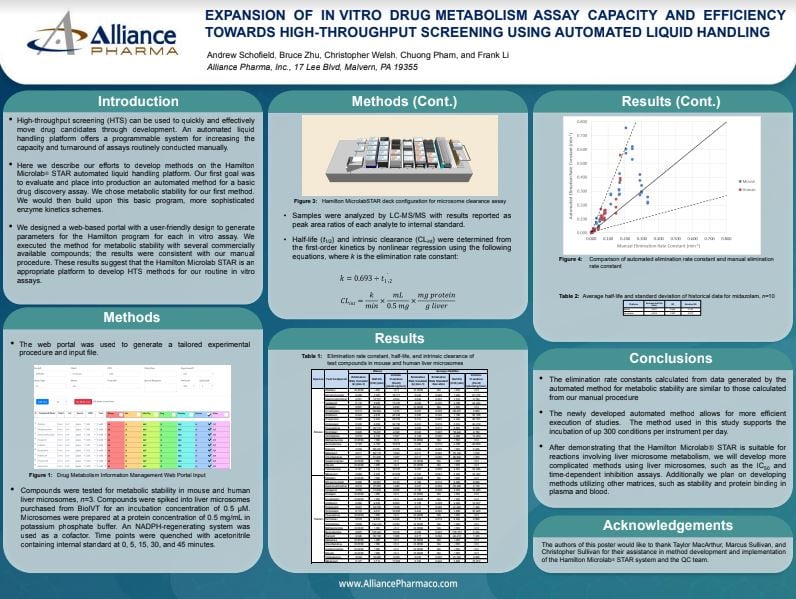
Explore the history that shaped Resolian and get a sneak peek into our future plans with our new video series.
Explore the history that shaped Resolian and get a sneak peek into our future plans with our new video series.
In the series of nine videos our CEO, Patrick Bennett explains the journey behind our recent rebranding and future plans in an insightful interview with Chris O'Connor from Life Science Marketing Radio.
Chapter 1 - "We Are Resolian"
Discover the journey behind our recent rebranding directly from our CEO, Patrick Bennett, in an insightful interview with Chris O'Connor from Life Science Marketing Radio.
Get ready for an exclusive look into the heart of Resolian, as we share our resolution to be one of the best laboratories globally committed to our purpose and mission in serving customers and our scientific staff.
🎬 Watch the video here:
Chapter 2 - "One Global Organization"
Our journey into becoming a unified global entity in just 15 months has set the stage for unparalleled flexibility and efficiency. The decision to consolidate under one name allows the organization to function seamlessly as a single entity, providing clients with the flexibility to manage clinical trials across the strategically located laboratories in the US, Europe, China, and Australia.
🎬 Watch the video below.
Chapter 3 - "Empowering Global Collaboration"
The acquisition of China-based Denali Medpharma in November 2023 marks a significant leap forward in our global capabilities, particularly benefiting clients navigating the complexities of the Chinese market.
With a team of high-quality scientists, the lab brings valuable expertise, bridging the gap for companies unfamiliar with the intricacies of operations in China. Our unique advantage lies in seamless method transfers between Western pharma clients and the China lab.
🎬 Watch the video here to find out more about what the new lab in China means for our clients.
Read more about the Resolian acquisition of Denali Medpharma here:
Chapter 4 - "Australia Lab - Accelerating Success"
The Australia lab emerges as a game-changer for clients seeking accelerated timelines and financial advantages in early-phase clinical studies. The regulatory landscape in Australia facilitates faster initiation of first-in-human studies compared to the US and Europe, supported by a network of robust clinical facilities.
Beyond speed, there is also a financial benefit to conducting trials in Australia.
Companies can claim 43.5% R&D cash refund with the Australian Government clinical trial rebate program, delivering a dual advantage in both efficiency and finance.
🎬 Watch the video here to find out more about Resolian’s Australian lab.
Chapter 5 - "Logistics Mastery for Seamless Global Operations"
Handling samples across borders involves intricate considerations, from stability to hazards. Resolian's expertise shines in navigating these complexities, particularly evident in China where HGRAC regulations pose challenges to the movement of human-based samples.
🎬 Watch the video here to find out more about Resolian’s logistics capabilities.
Chapter 6 - "Tailored Solutions for Sponsors"
Sponsors often manage a portfolio of vendors. Most want to have some larger full-service companies in their portfolio of bioanalytical vendors. Depending on the project, those full-service vendors aren’t always the right fit. Most of them are not global. A sponsor might take advantage of one component of their service but are not necessarily what you need for this specific project.
There is always a need for laboratories like Resolian that are focused on bioanalysis.
🎬 Watch the video here to find out more about Resolian’s responsiveness and flexibility.
Chapter 7 - "Diverse Analytical Prowess Unveiled"
Unlocking the depths of Resolian's analytical capabilities reveals a remarkable breadth across various specialties. While many labs excel in specific areas, each of Resolian's entities stands out for its comprehensive strength. With extensive experience ranging from immunoassay to LCMS, including small molecules, large molecules, biologics, and oligonucleotides, Resolian covers a spectrum of analytical domains.
🎬 Watch the video here to find out more about the diverse analytical skills at Resolian.
Chapter 8 - "Balancing Unity and Uniqueness"
Four bioanalytical labs, four continents.
Navigating the question of maintaining consistency, Resolian unveils a dual approach to harmonize operations seamlessly. While integration is implemented for non-scientific functions, ensuring universal policies with local modifications, the scientific operations follow a unique path of harmonization.
🎬 Watch the video here to find out more about our goal to create a familiar environment for legacy customers, facilitating flexibility between geographies.
Chapter 9 - "Unveiling the Heartbeat - Bringing Benefits to Patients!”
In this final instalment, we explore Resolian's impact on patients' experiences in clinical trials. Unlike central labs or clinical diagnostics, bioanalysis often involves unique methods, limiting optimal locations for sample analysis.
Resolian's global network, with laboratories in multiple geographies, provides our pharma clients the leverage to strategically choose where they run analyses in alignment with their clinical trials. This not only expands the options for clients but, more importantly, broadens the reach of clinical trials, benefiting a larger population of patients around the globe.